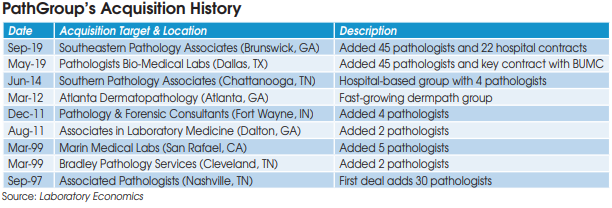
Originally founded by pathologists in 1965, PathGroup (Brentwood, TN) has grown to become the nation’s largest privately-held lab company. PathGroup, which currently has 2,200 employees, is owned by Pritzker Private Capital, company management and pathologists. Laboratory
Economics recently spoke with long-time Chief Executive Ben Davis, MD, to discuss PathGroup and its growing role in Covid-19 testing.
When did PathGroup initiate Covid-19 PCR testing and how many are being performed?
We began Covid-19 PCR testing on April 1st with an initial capacity of 2,000 tests per day using the Hologic Panther and Roche cobas 6800 platforms. We are currently performing an average of about 7,500 tests per day with capacity of up to 14,000 tests per day. Demand is coming from physician clinics, state health departments, employers, schools, universities, hospitals and nursing homes. Peak demand came in early July when we received specimens in excess of our capacity for several days.
How have positivity rates trended?
On July 6 our positivity rate peaked at 17%. That’s dropped to a current average of about 10%, including 12.5% for patients under age 30 and 9% for those 30 and above.
What is your turnaround time for Covid-19 PCR testing?
Our turnaround time from specimen collection to result reporting is consistently 24 hours for all clients. We have limited new client additions in order to maintain a 24-hour turnaround time. The location of our main 150,000-square-foot lab—adjacent to Nashville International Airport—has also helped.
Could PathGroup do more Covid-19 PCR tests if it had more supplies?
Yes. The current shortages of specimen collection devices and test kits are keeping us below instrument capacity. We’ve kept in close touch with our suppliers on changing situations and plan accordingly.
Are most private insurers matching Medicare’s $100 reimbursement rate for Covid-19 PCR testing?
Yes.
Describe the NIH grant that PathGroup was awarded and how it will be used?
PathGroup has received a $20.75 million grant from the National Institutes of Health (NIH) under its Rapid Acceleration of Diagnostics (RADx) program. The grant funding will be used to purchase new high-throughput liquid handling, robot and automated testing equipment from Illumina, LGC, Hologic and Thermo Fisher. We also plan to hire an additional 100 to 200 employees with the goal of expanding our Covid-19 PCR testing capacity to 80,000 tests per day. Importantly, we’ll be diversifying our supply chain to mitigate risk against supply chain constraints as fall/winter approaches.
Which Covid-19 antibody test does PathGroup perform?
Roche’s cobas serum antibody test. We’re performing about 400 tests per day.
Have non-Covid-19 clinical and pathology test volumes bounced back from the lows?
Yes. In late March/early April, our volumes had declined temporarily by 75% with anatomic pathology case volume hit the hardest. But in May, as physician offices started re-opening and elective surgeries resumed, we saw a sharp rise in non-Covid-19 volumes that are currently back at 100% of pre-pandemic levels, maybe even a little higher.
What precautions have you taken for your employees?
About 15% to 20% of our 2,200 employees are currently working from home. All people entering our lab facilities have their temperature checked and must wear masks. We are also offering Covid-19 testing on a voluntary basis to any employee that requests one, whether symptomatic or asymptomatic.
Do you think Covid-19 will come back in a severe way this fall/winter?
I would not want to predict severity, but the disease is likely to continue to spread this coming fall/winter. A vaccine will help, but Covid-19 is not going away. One of my biggest fears is that the disease will adapt and mutate into a more virulent strain, such as SARS-CoV-1 in 2003 and the MERS-CoV in 2012.