Why Does Big Pharma Support LDT Regulation?
Why Does Big Pharma Support LDT Regulation?
Friends of Cancer Research (FOCR-Washington, DC) has been a steadfast advocate for FDA regulation of LDTs (both the VALID Act and the FDA’s final rule). FOCR contends that FDA regulation will reduce variability in diagnostic tests used to identify cancer patients who are most likely to benefit from cancer therapy.
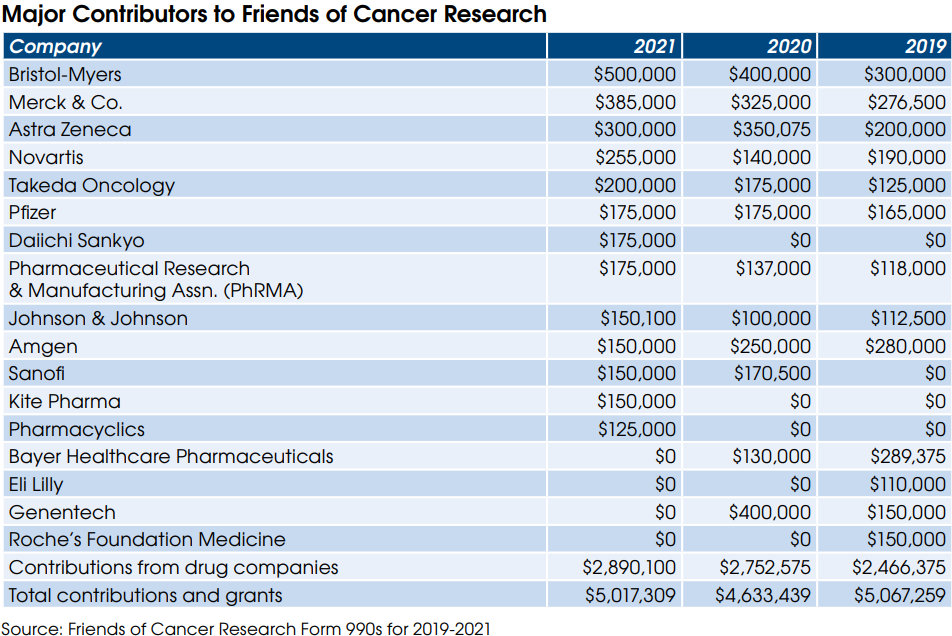
The nonprofit FOCR has a mission “to accelerate policy change, support groundbreaking science, and deliver new therapies to patients quickly and safely.” A Laboratory Economics’ analysis of FOCR’s Form 990 tax statements shows that it gets most of its funding from major pharmaceutical companies.
Twelve pharmaceutical companies and their trade organization, the Pharmaceutical Research & Manufacturing Assn. (PhRMA), contributed a total of $2.9 million to FOCR in 2021, according to FOCR’s latest publicly available Form 990 tax statement. This accounted for 58% of the total contributions and grants received by FOCR in 2021.
Over the three-year period (2019-2021), the biggest contributors to FOCR have included BristolMyers ($1.2 million), Merck ($986,500) and Astra Zeneca ($850,075). PhRMA also contributed $430,000.
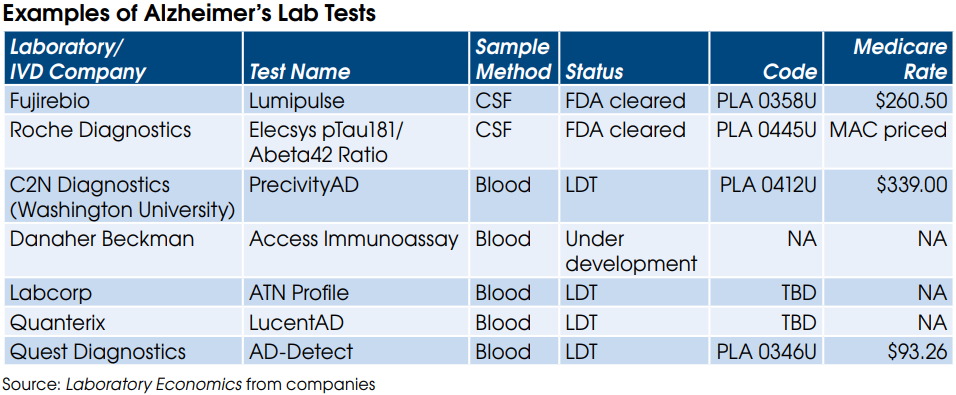
The pharmaceutical companies supporting FOCR are developing expensive oncology drugs targeted at cancer patients with specific genetic mutations. For example, in November 2023, the FDA approved Bristol-Myers’ Augtyro to treat adult patients with locally advanced or metastatic ROS1-positive non-small cell lung cancer. The wholesale price for Augtyro is $29,000 per month/$348,000 per year. There is no FDA-cleared test to detect ROS1 rearrangements for selecting patients for treatment with Augtyro. LDT test panels for ROS1 include LungOI (PLA 0414U; Medicare rate: $706) performed by the Imagene Lab (Phoenix, AZ).
Laboratory Economics thinks that big pharma may be concerned that some LDTs have erroneously high cut-off values—the benchmarks that determine a positive or negative test result—that can limit patient access to cancer drugs.
FOCR President Jeff Allen, PhD, gave testimony on LDTs before the U.S. House of Representatives’ Committee on Energy and Commerce, Subcommittee on Health on March 21, 2024. Allen said that FDA regulation of LDTs was necessary to “Establish uniform performance standards, regulatory processes, and transparency for all diagnostic tests to ensure accuracy of results.”